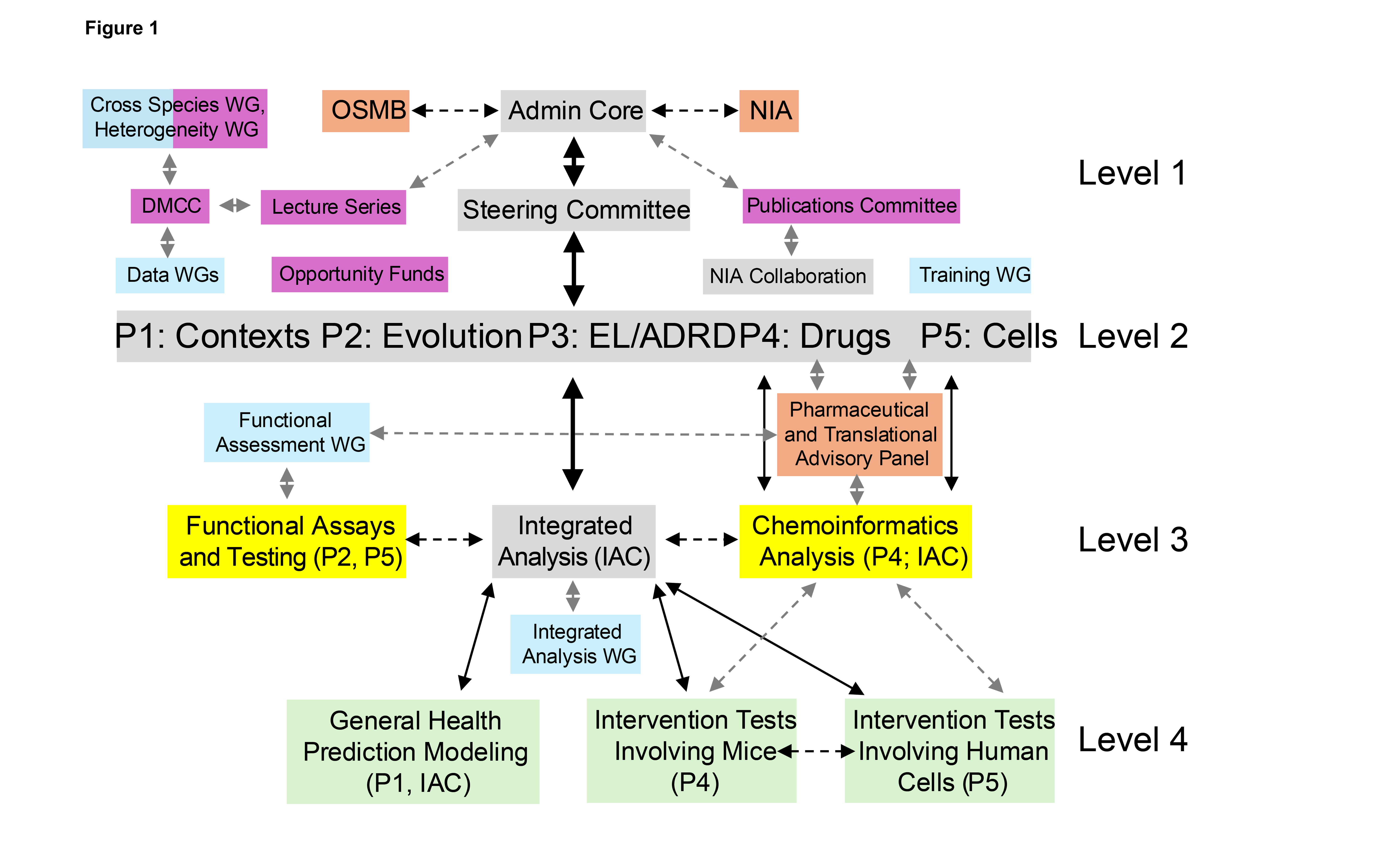

There are three advisory and oversight components, color coded in orange, and these include the Observational Study Monitoring Board (OSMB), the NIA program office, and the Pharmaceutical and Translational Advisory Panel (PTAP). The OSMB and NIA participate in aspects of the overall decision-making for the LC, whereas the PTAP focuses on directions in drug and general product development. To enable and enhance the scientific activities of the consortium and enhance internal collaborations, five working groups (WGs) were established (color coded light blue in Figure 1): a ‘training WG’ focusing on pre- and post-doctoral researchers as well as funded career development awardees (e.g., K-Awardees) working with LC investigators; a ‘functional assessment WG’ pursuing studies of the functional effects of analytes found to be associated with longevity and healthy aging; two data sharing WGs: one focused on general data set access, storage and use and one focused on data sets with restrictions on their use; and an ‘Integrated Analysis (IA)’ that considers findings from the consortium as a whole that might benefit from replication, integration and triangulation.
The LC also is heavily involved in the operation of two broader working groups overseen by the DMCC: a ‘cross species WG’ and a ‘heterogeneity analysis WG’. These WGs involve participants across all the ELITE consortia. Four administrative components help with the logistics, coordination and outreach of the LC operations (color coded in purple) and include a publications and presentations committee, an opportunity funds mechanism to support research that expands the consortium activities, a lecture series that provides exposure to the consortium, and routine interactions with the DMCC for issues of data access, governance, and deposits. Two integrated activities that involve direct cross project and core interactions are color coded in yellow and include carrying out functional assays on analytes found to be associated with longevity and healthy aging across the consortium and refining lists of candidate drugs and interventions for broader development. Finally, three products and deliverables are color coded in light green and include the development of clinically meaningful prediction models, the identification of candidate drug/intervention targets, and the identification of candidate compounds for testing in pre-clinical models. In addition, the different levels of communication and information flow across the consortia components and researchers are provided in the right-hand column of Figure 1. These levels correspond to: 1. Administrative oversight and governance; 2. Day-to-day scientific interactions within and between the projects; 3. Focused integration of results and data; and 4. Considerations involving preclinical product development that emerges from the research.