Overview
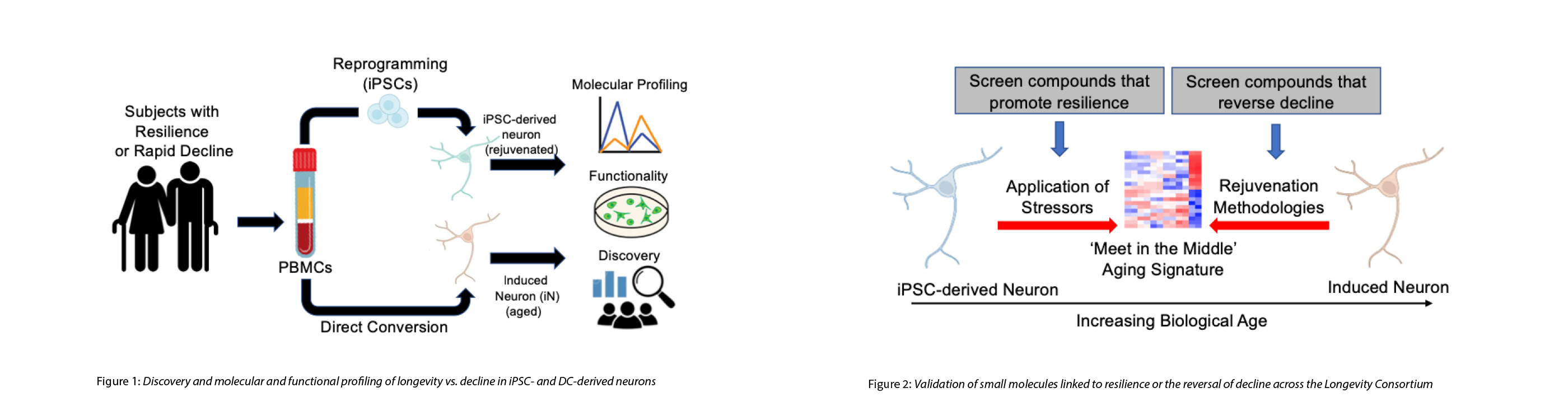
The mechanisms that drive resiliency or, conversely, rapid decline, remain unclear. Models of human aging, longevity, and resilience to disease that allow for the functional testing of potential interventions are virtually non-existent. To directly address these gaps in understanding, we are building and harnessing novel, complementary, in vitro human cellular models created from the fibroblasts and/or Peripheral Blood Mononuclear Cells (PBMCs) of healthy young and old subjects and those experiencing cognitive impairment. First (1), in combinatorial efforts, fibroblasts and PBMCs are being reprogrammed into iPSCs and differentiated into neurons (iPSC-derived neurons) while, in parallel, the same starting material is undergoing direct conversion into induced neurons (iNs) which retain aging-associated signatures. Using this approach, we are harnessing both the flexibility and unlimited cell source provided by iPSCs while also capturing potential epigenetic drivers of longevity or resilience by using iNs. Next (2), we are exploiting these complementary cell-based models to identify mechanisms underlying prevention (iPSC model) or reversal (iN model) of aging-related decline including studies and models of dynamic resilience, DNA repair hotspots, and metabolic functionality. Lastly (3), we will leverage iPSCs and iNs in functional validation studies of the genes, genetic variants, proteins, metabolites, and other analytes found to be associated with longevity or decline across all projects in the Longevity Consortium. These overlapping strategies present a unique opportunity for the cross validation of the functional results, identified pathways, and signatures observed across systems and laboratories, a major point of concern in the rapidly emerging geroscience field.

Leadership
Key Personnel
Salk Institute
Adler Professor at the Salk Institute, Adjunct Professor, UCSD and past President of Salk. Dr. Gage received his Ph.D. from The Johns Hopkins University. His work concentrates on the adult central nervous system and unexpected plasticity and adaptability to environmental stimulation. He models human neurological and psychiatric disease in vitro using human stem cells, and also studies the genomic mosaicism that exists in the brain as a result of mobile elements active during neurogenesis.
Boston University
Associate Professor in the Department of Medicine at Boston University School of Medicine, Director of Research in the Division of Hematology-Oncology at Boston Medical Center. Dr. Murphy is also the co-founder of the Boston University and Boston Medical Center’s Center for Regenerative Medicine (CReM) (www.bumc.bu.edu/stemcells), where 70 scientists including 12 faculty members work together, synergistically in a multi-disciplinary approach to advancing stem cell biology and regenerative
UC Riverside
Thomas Girke is a Professor of Bioinformatics at the Institute for Integrative Genome Biology at the University of California, Riverside (UCR). He holds positions as Director of the High-Performance Computing Center (HPCC) and Director of the Graduate Program in Genetics, Genomics and Bioinformatics (GGB). His research interests are in the areas of cheminformatics and bioinformatics, with a focus on both discovery and methodology development.
Team Members